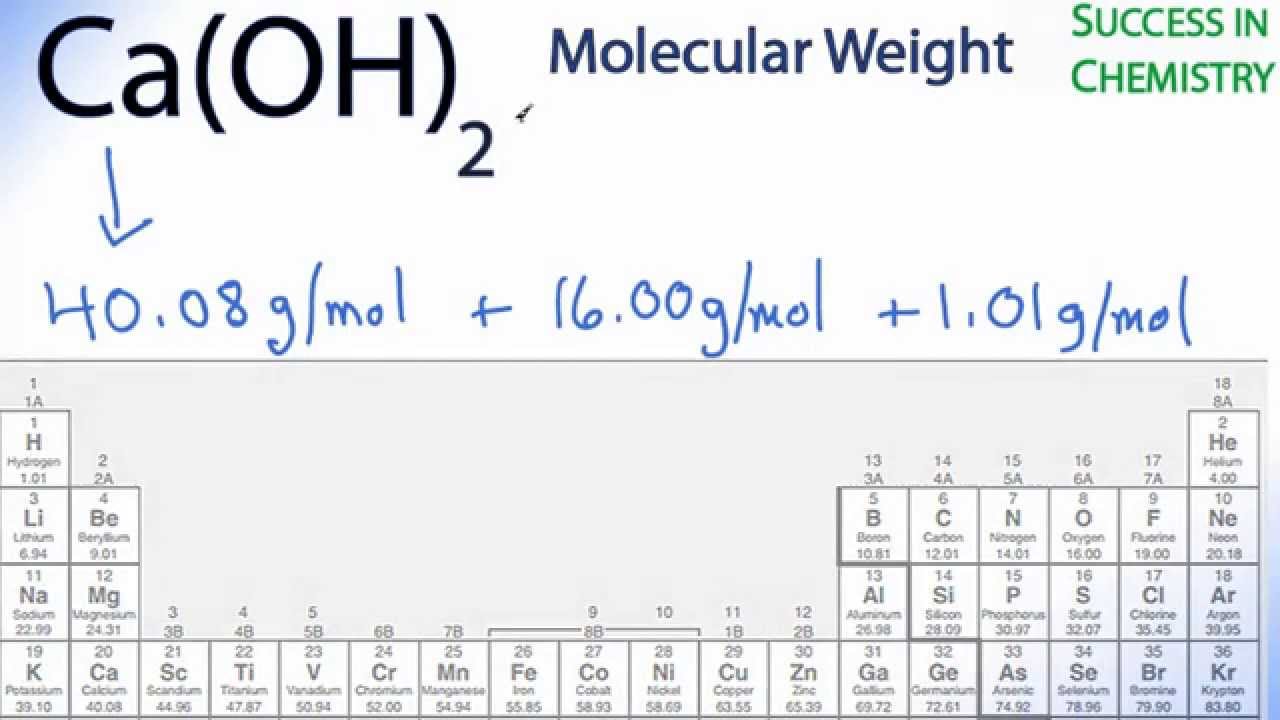
It is the fifth most abundant element in Earth's crust, and the third most abundant metal, after iron and aluminium. Its physical and chemical properties are most similar to its heavier homologues strontium and barium. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Compute Mass of Each Element.Calcium is a chemical element with the symbol Ca and atomic number 20. In practice this means gases with densities approximately corresponding to I FjfSUrc~ in 1.,:,(lckc.t3 indico.tc the literature TG£c.nmcc" At the end of S-c ctioo. 29.45/100 x 25.6.such that the rates of transport of mass, momentum, and l energy are entirely controlled by binary molecular collisions. To calculate the mass of Ca present in a 25.6-g sample of this compound, the correct procedure is given by. Converting to moles gives 6.65 mol of C and 19.9 mol of H. 100.0g of the compound contains 79.9g of C and 20.1g of H. Convert molar mass to density using the conversion factor above: In most literature you will find density in units of g/mL or g/cm 3 so to convert from g/L to g/mL or g/cm 3. Molar mass (SO3) = atomic mass oxygen x 3 + atomic mass sulfur = 3 (15.99 g/mol) + 32.06 g/mol = 80.066 g/mol SO3. The first step to finding the molar mass of Nickel(Ii) Dithionate is to count the number of each atom present in a single molecule using the chemical formula, Ni(SO3)2: Element Number of Atoms Molar mass of NaCl is 58.443, how many grams is 5 mole NaCl? Molar mass of AgNO3 is 169.873, 2 kg AgNO3 is equal to. The mass (in grams) of a compound is equal to its molarity (in moles) multiply its molar mass: grams = mole × molar mass. Cupric sulfate is used primarily for agricultural purposes, as a pesticide, germicide, feed additive, and soil additive.The mass and molarity of chemical compounds can be calculated based on the molar mass of the compound. 200 grams SO3 to mol = 2.49803 mol.Cupric sulfate is a salt created by treating cupric oxide with sulfuric acid.This forms as large, bright blue crystals containing five molecules of water (CuSO4∙5H2O) and is also known as blue vitriol.The anhydrous salt is created by heating the hydrate to 150 ☌ (300 ☏). Determine the molar mass for the following. 1.32 moles Si 7.29 mol CO2 712.4 g AuCl Molar Mass worksheet KEY. Determine how many atoms/molecules in the following samples. 
Determine the number of moles in the following samples. The given mass of K (4.7 g) is a bit more than one-tenth the molar mass (39.10 g), so a reasonable "ballpark" estimate of the number of moles would be slightly greater than 0.1 mol.Determine the mass of the following samples. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. SO3 H2O = H2SO4 in the reaction with water, while releasing a lot of heat, and. The atomic weight of gold is 196.97 grams per mole.Molar Mass: 80.06: Density: 1.97g/mLat 25☌(lit.) Melting Point: 16.8☌(lit.). A physicist wants to know the mass of 1.8 x 10^22 atoms of gold. Using the atoms to grams calculator, the chemist determines that the mass of the carbon is 4.17 grams. The atomic weight of carbon is 12.01 grams per mole. The most common example is the molar volume of a gas at STP (Standard Temperature and Pressure), which is equal to 22.4 L for 1 mole of any ideal gas at a temperature equal to 273.15 K and a pressure equal to 1.00 atm.Aprende a hallar la MASA MOLECULAR o MASA MOLAR (también llamadas PESO MOLECULAR o MOLAR) fácil en 5 SENCILLOS PASOS.⏰ TIEMPOS:00:00 Introducción00:18 ¿Molec.Explanation of how to find the molar mass of Cr2(SO4)3: Chromium (III) sulfate.A few things to consider when finding the molar mass for Cr2(SO4)3:- make sure.A chemist wants to know the mass of 2.5 x 10^24 atoms of carbon. Compound.The molar volume of a gas expresses the volume occupied by 1 mole of that respective gas under certain temperature and pressure conditions. Choose Compound StatesThe characteristic molar mass of an element is simply the atomic mass in \(\frac weight and moles. Thermodynamics of the reaction can be calculated using a lookup table. Sulfuric Acid - H 2 SO 4 Oil Of Vitriol Hydrogen Sulfate Battery Acid Sulphuric Acid H2So4. Molar mass of SO3 (Sulfur trioxide) is 80.0632 g/mol Convert between SO3 weight and moles Elemental composition of SO3 Computing molar mass step by step First, compute the number of each atom in SO 3: S: 1, O: 3 Then, lookup atomic weights for each element in periodic table: S: 32.065, O: 15.9994SO3 Molar Mass SO3 Oxidation Number.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
